
As a newer pharmaceutical dosage form, effervescent tablets are different from ordinary tablets that the former uses the reaction of organic acid and basic carbonate (hydrogen) salt as an effervescent disintegrant. When placed in water, an effervescent reaction occurs immediately. Generate and release a large amount of carbon dioxide gas like boiling, hence the name effervescent tablet.
What Are the Ingredients and Examples of Effervescent Tablet?
Oral effervescent tablets are mainly divided into two categories: medicinal and health care. The medicinal type is mainly used to treat colds, infections and other diseases, such as aspirin effervescent tablets, acetaminophen effervescent tablets and acetylcysteine effervescent tablets. Health care effervescent tablets mainly contain vitamins and minerals, such as vitamin C effervescent tablets, multidimensional effervescent tablets, calcium supplement effervescent tablets and iron supplement effervescent tablets.
Topical effervescent tablets are generally used for medicinal purposes and are mainly used to treat gynecological inflammation. Effervescent tablets are used in gynecological local medication. They have the characteristics of fast onset, complete absorption and no injury to mucosa. Their dissolution rate is faster than suppository and their absorption is better than suppository.
The prescription of effervescent tablets consists of main drug, diluent, adhesive, disintegrating agent, lubricant and other excipients. The types of diluent, adhesive, lubricant and other excipients used are the same as those of ordinary tablets. Only the appropriate varieties need to be selected according to the preparation process.
Different from ordinary tablets, the disintegrating agent used in effervescent tablets is effervescent disintegrating agent. Effervescent disintegrating agent includes acid source and alkali source. Common acid sources include citric acid, malic acid, boric acid, tartaric acid, fumaric acid, inorganic mineral acid (hydrochloric acid), etc. Common alkali sources include sodium bicarbonate, sodium carbonate and their mixtures. The acid-base ratio has a significant impact on the preparation and stability of effervescent tablets. It is generally believed that the amount of acid exceeds the theoretical amount, which is conducive to the stability of effervescent tablets and improve the taste.
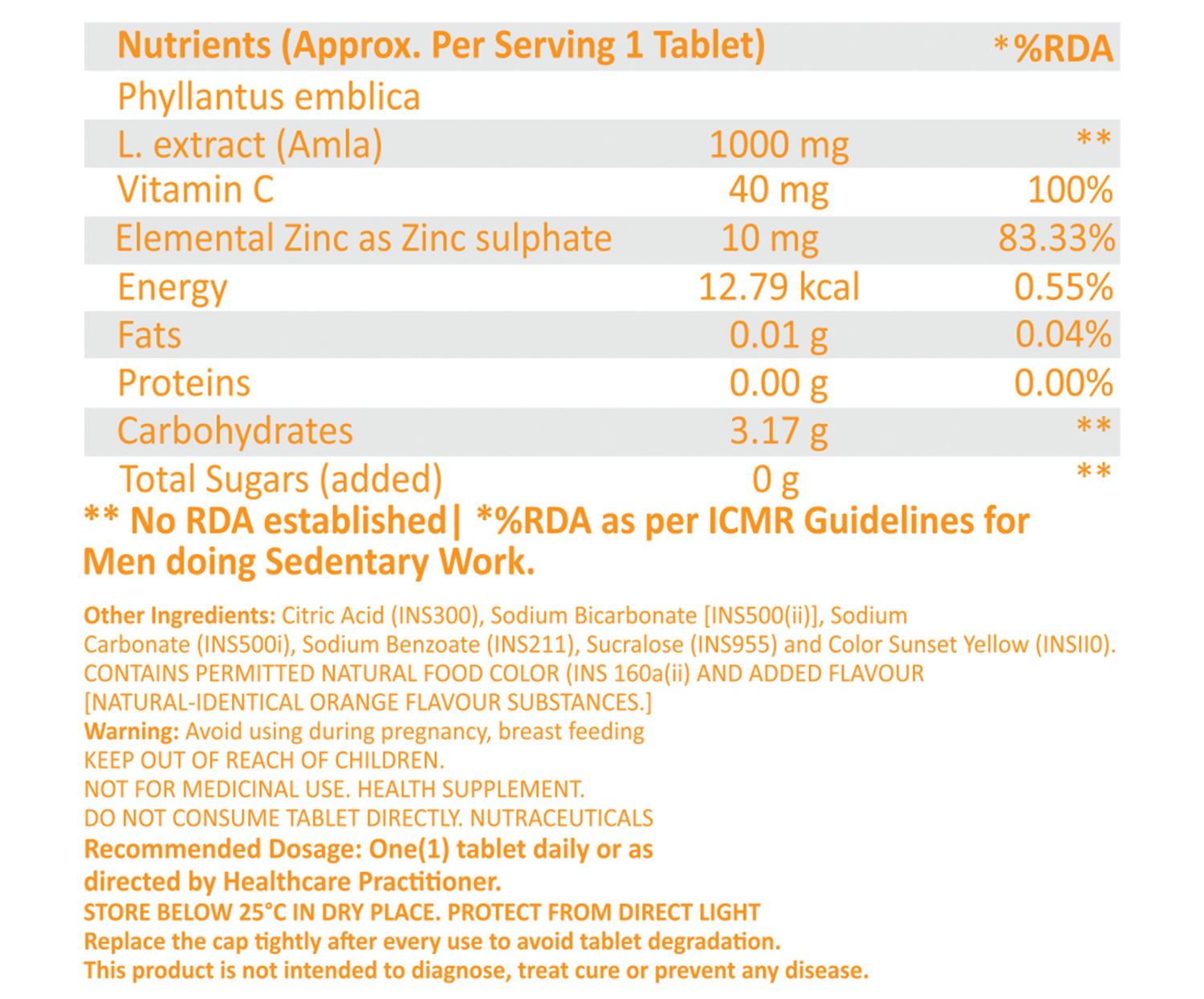
Example of Effervescent Tablet Ingredients
How Are Effervescent Tablets Manufactured?
At present, with the development of pharmaceutical technology, the conventional preparation methods of effervescent tablets are mainly divided into three types: wet granulation with tablet pressing, dry granulation with tablet pressing and direct tablet press.
One of the purposes of granulation is to ensure that the material can flow into the mold hole in the state of particles with good fluidity, so as to form the tablet; Second, after the granules are made, the pressure requirement is smaller than that of the powder. Because the surface of the granules is uneven, it has the function of "embedding each other", which can overcome the quality problems such as the looseness and cracking of the sugar tablets; Third, solve the quality problems such as easy sticking and punching of fine powder, and avoid the flying of fine powder when the powder is directly pressed.
Except that some crystalline raw and auxiliary materials can be directly pressed into tablets, powdered raw and auxiliary materials are generally pressed into tablets after granulation.
Wet Granulation and Tablet Pressing
When the adhesive is an aqueous solution, in order to avoid acid-base reaction during granulation, it is advisable to separate the acid source and alkali source of effervescent disintegrating agent for granulation, drying, mixing evenly and tablet pressing. Theoretically, the use of anhydrous ethanol and other organic solvents for granulation is conducive to the stability of the preparation, but it is difficult to ensure that they are completely anhydrous, which may affect the stability of the preparation and increase the cost.
Adjust the weight of the produced varieties to make them consistent; Then feeding, tablet pressing and pushing, and the semi-automatic or full-automatic tablet pressing machine is used to complete the tablet pressing process.
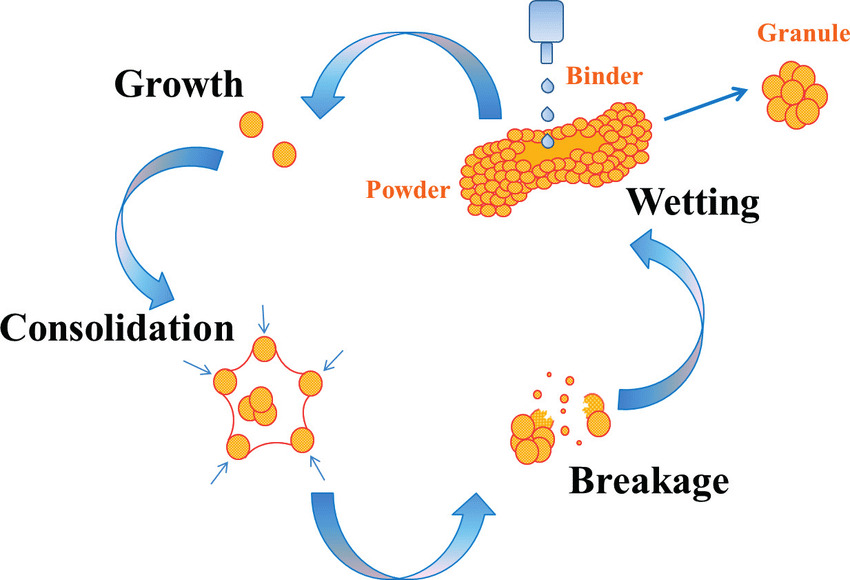
SOP of Wet Granulation
Tablet Pressing After Dry Granulation
Dry granulation can be operated continuously, with low energy consumption and high output. The biggest advantage is that there is no need to add adhesive in the granulation process, thus avoiding the contact between the acid source and alkali source of effervescent disintegrating agent and water to the greatest extent, which is very conducive to improving the stability of effervescent tablets.
The dry granulation method generally adopts the rolling method. After the materials are mixed evenly, they are rolled evenly by the roller (double drum rubber mixer) and pressed into thin sheets with the required hardness and thickness. They enter the granulation machine, add lubricant, and then are mixed by the V-shaped mixer assembly, and then they can be formed by using the tablet press.
Direct Powder Tablet Pressing
Select appropriate drug components and excipients, and directly press tablets without granulation. It has the advantages of time saving, energy saving, simple process, avoiding contact with water and increasing the stability of effervescent tablets. However, this method requires high fluidity and compression formability of materials, so it is limited in practical application.
In addition to powder direct tablet pressing, there is also crystallization direct tablet pressing process. Some crystalline granular raw materials have good fluidity and compressibility, and many materials containing crystalline water can be directly pressed by adding an appropriate amount of lubricant. However, it is easy to burst during tablet pressing, so inspection shall be paid attention to.
What's the Difference between Effervescent Tablets and Normal Tablets?
From taking to absorption, drugs generally need to undergo the process of disintegration, dissolution and absorption by the human body. The following two aspects will explain the difference between the two.
Disintegration and Dissolution Process
The tablets are quickly disintegrated after being put into water outside the body, releasing a large amount of carbon dioxide gas and dissolving into liquid. Generally, the disintegration time limit is 1-5 minutes. The common tablet after oral administration, the common tablet slowly disintegrates and dissolves in the gastrointestinal tract, and the drug is released. The disintegration time limit of different tablets is 15~60 minutes.
Absorption and Effect
The effervescent tablets disintegrate and dissolve the drug in vitro. After oral administration, the liquid is widely distributed in the gastrointestinal tract and absorbed into the blood through the gastrointestinal tract for about 10~30 minutes, which reduces the local irritation of the drug in the gastrointestinal tract, thus reducing the side effects and achieving a quick and efficient effect. After oral administration, the common tablet disintegrates in the stomach, the drug slowly dissolves out and is absorbed into the blood through the gastrointestinal tract. It takes 2~3 hours to reach the effective blood drug concentration, and the effect is slow.
Normal Tablets VS Effervescent Tablets
What Are the Precautions When Taking Effervescent Tablets?
- Do not take tea or drink to prevent chemical reaction.
- Children should take it under the supervision of their parents.
- Vitamin C effervescent tablets should be drunk and foamed immediately, and vitamin C oxidation fails after being placed for too long.
- When using effervescent tablets, the water temperature should not be too high, only about 40 ℃, especially vitamin C effervescent tablets. Too high water temperature will seriously affect the efficacy.
- After taking vitamin C effervescent tablets, rinse your mouth with clean water to reduce the stimulation of acid substances on teeth and avoid tooth damage.
- Effervescent tablets contain a lot of sodium. Taking them for a long time will increase the risk of cardiovascular and cerebrovascular diseases. Therefore, those with high blood lipid and blood pressure, family history or other risk factors of cardiovascular and cerebrovascular diseases should use effervescent tablets less or with caution. Elderly people who often use effervescent tablets should pay attention to monitoring their blood pressure, and patients with hypertension should appropriately reduce their salt intake during taking.
How to Protect Effervescent Tablets Properly?
Production Control Environment and Packaging Conditions
In the packaging process, it is necessary to control the process prescription, as well as the temperature and humidity of the environment, so as to avoid the bulging of aluminum foil due to component reaction. The temperature and humidity of the production environment from granulation to effervescent tablet packaging shall be strictly controlled. Generally, the temperature shall be 18-20 ℃, and the humidity shall be less than 45%. The time interval from granulation to packaging shall be as short as possible.
The packaging of effervescent tablets is generally completed by the tablet tube filling machine of professional manufacturers. The parts in contact with the products are made of 316 stainless steel, which meets the medical grade standard.
Daily Storage
Effervescent tablets need to be placed in a cool and dry place away from water and light. For example, the common vitamin C effervescent tablets have strong reducibility, fast light decomposition, easy oxidation, and are not heat-resistant. Therefore, they need to be placed in a cool and dry place away from water and light, otherwise they are easy to deteriorate.





 Wechat
Wechat




